
FILGRASTIM PATIENT INFORMATION BOOKLET
1. PATIENT GUIDE:
Back to top- Keep this leaflet as you may need to read it again.
- If you have further questions, please ask your doctor or your pharmacist.
- FILGRASTIM TEVA has been prescribed for you personally and you should not share your medicine with other people. It may harm them, even if their symptoms are the same as yours.
2. NEUTROPENIA
Back to topNeutropenia is an abnormally low level of neutrophils. Neutrophils are a common type of white blood cell important for fighting off infections — particularly those caused by bacteria.
Cancer chemotherapy is probably the most common cause of neutropenia. People with chemotherapy-related neutropenia are prone to infections while they wait for their cell counts to recover.
Neutrophils are manufactured in bone marrow — the spongy tissue inside some of your larger bones. Anything that disrupts neutrophil production can result in neutropenia2
SIGNS AND SYMPTOMS OF NEUTROPENIA
Back to topNeutropenia itself may not cause any symptoms. Patients usually find out they have neutropenia from a blood test or when they get an infection. Some people will feel more tired when they have neutropenia. Your doctor will schedule regular blood tests to look for neutropenia and other blood-related side effects of chemotherapy.
For patients with neutropenia, even a minor infection can quickly become serious. Talk with your health care team right away if you have any of these signs of infection: 4
- A fever, which is a temperature of 38 °C or higher
- Chills or sweating
- Sore throat, sores in the mouth, or a toothache
- Abdominal pain
- Pain near the anus
- Pain or burning when urinating, or urinating often
- Diarrhoea or sores around the anus
- A cough or shortness of breath
- Any redness, swelling, or pain (especially around a cut, wound, or catheter)
- Unusual vaginal discharge or itching
3. WHAT YOU NEED TO KNOW BEFORE YOU USE FILGRASTIM TEVA?
Back to topDo not use FILGRASTIM TEVA:
- If you are hypersensitive (allergic) to filgrastim or any of the other ingredients of FILGRASTIM TEVA.
- If you have severely low white blood cell levels that were detected soon after your birth (Kostmann’s syndrome) with abnormal cell structure or function.
- If you have kidney or liver problems.
- If you are pregnant or breastfeeding your baby.
Tell your doctor or healthcare professional before being given the injection if:
- You have a history of sickle cell disease because FILGRASTIM TEVA can cause sickle cell crisis (a painful episode that occurs in people who have sickle cell anaemia. Sickle-shaped red blood cells block blood vessels. Because of this, blood and oxygen cannot get to the tissues, causing pain).
- If you have osteoporosis (bone disease). Your doctor will monitor your 5 bone mineral density if you are receiving FILGRASTIM TEVA for more than six months.
TAKE SPECIAL CARE WITH FILGRASTIM TEVA IF:
Back to topYou have symptoms of cough, fever, shortness of breath or trouble breathing because these may be signs of a serious lung problem called Acute Respiratory Distress Syndrome (ARDS). Stop treatment with FILGRASTIM TEVA immediately and consult your doctor if you have any of these symptoms.
4. POSSIBLE SIDE EFFECTS OF FILGRASTIM TEVA
Back to topFILGRASTIM TEVA can have side effects.
Should your general health worsen or if you experience any untoward effects while taking this medicine, please consult your doctor, pharmacist or other healthcare professional for advice.
If any of the following happens, stop taking FILGRASTIM TEVA and tell your doctor immediately or go to the casualty department at your nearest hospital:
- Weakness, drop in blood pressure, difficulty breathing, swelling of the face (anaphylaxis)
- Skin rash, itchy rash (urticaria)
- Swelling of the face, lips, mouth, tongue or throat (angioedema)
- Shortness of breath (dyspnoea) These are all very serious side effects. If you have them, you may have had a serious allergic reaction to FILGRASTIM TEVA. You may need urgent medical attention or hospitalisation. Tell your doctor immediately or go to the casualty department at your nearest hospital if you notice any of the following: 6
- Cough, fever and difficulty breathing as this can be a sign of Acute Respiratory Distress Syndrome (ARDS)
- Severe pain in the bones, chest, gut or joints (sickle cell crisis)
These are all serious side effects. You may need urgent medical attention
SIDE EFFECTS THAT MAY OCCUR FREQUENTLY
Back to top- Nausea (feeling sick)
- Vomiting
- Constipation
- Diarrhoea
- Lack of appetite
- Headache
- Sore throat
- Extreme tiredness (fatigue), general weakness
- Hair loss
- Chest pain
- Musculoskeletal pain
Information about other less frequently occurring side effects are included in the package insert enclosed in the FILGRASTIM TEVA carton.
If you notice any side effects not mentioned in this leaflet, please inform your doctor or pharmacist.
5. INFORMATION FOR INJECTING YOURSELF
Back to topIf you are not sure about giving yourself the injection or you have any questions, please ask your doctor or nurse for help.
HOW DO I INJECT FILGRASTIM TEVA MYSELF?
Back to topFILGRASTIM TEVA is supplied in packs of 5 pre-filled syringes each fitted with a safety device.
FILGRASTIM TEVA should be stored between 2º C – 8º C in a refrigerator.
For a more comfortable injection, take your FILGRASTIM TEVA pre-filled syringe out of the refrigerator and let the pre-filled syringe stand for 30 minutes to reach room temperature or hold the pre-filled syringe gently in your hand for a few minutes. Do not warm FILGRASTIM TEVA in any other way (for example, do not warm it in a microwave or in hot water).
Check the appearance of FILGRASTIM TEVA. It must be a clear and colourless liquid. If there are particles in it, you must not use it.
Do not remove the cover from the syringe until you are ready to inject.
Wash your hands thoroughly.
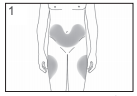
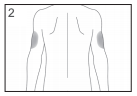
The most suitable places to inject yourself are:
- the top of your thighs; and
- the abdomen, except for the area around the navel.
Do not touch the needle or push the plunger
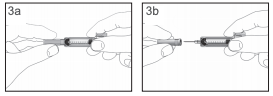
The syringe has a scale on the syringe barrel. Push the plunger up to the number (ml) on the syringe that matches the dose of FILGRASTIM TEVA that your doctor prescribed. Check again to make sure the correct dose of FILGRASTIM TEVA is in the syringe.
You can now use the pre-filled syringe
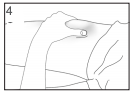
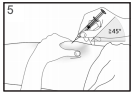
Inject the liquid slowly and evenly, always keeping your skin pinched.
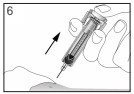
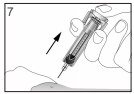
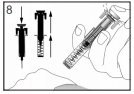
Only use each syringe for one injection. Do not use any FILGRASTIM TEVA that is left in the syringe
6. DISPOSING OF USED SYRINGES
Back to topPlease contact your healthcare treatment team with any questions that you may have.

- Package Insert (Patient information Leaflet) 27 November 2017.
- Neutropenia. Available at: https://www.mayoclinic.org/ symptoms/neutropenia/basics/definition/sym-20050854.
- Neutropenia. Available at: https://www.cancer.net/ navigating-cancer-care/side-effects/neutropenia.
Each pre-filled syringe contains 30 MIU (300 μg) or 48 MIU (480 μg) of filgrastim in 0,5 ml or 0,8 ml of solution for injection or infusion respectively. For full prescribing information please refer to the package insert approved by the medicines regulatory authority. TEVA PHARMACEUTICALS (PTY) LTD. Co. Reg. No. 1986/002796/07. Maxwell Office Park, Magwa Crescent West, Waterfall City, Midrand, 2090. Tel: +27 11 055 0200, Fax: +27 86 680 8988. Marketed by CIPLA MEDPRO (PTY) LTD. Co. Reg. No. 1995/004182/07. Building 9, Parc du Cap, Mispel Street, Bellville, 7530, RSA. Website: www.cipla.co.za Customer Care: 080 222 6662. [13602] ATC307/06/18 S

Filgrastim Teva – scripted patients only

Filgrastim Teva – scripted patients only

Filgrastim Teva – scripted patients only
Filgrastim Teva – scripted patients only

Filgrastim Teva – scripted patients only


